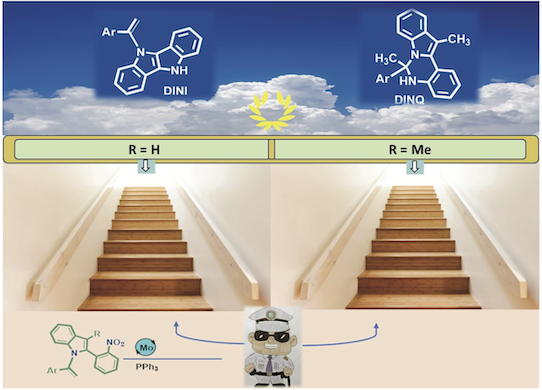
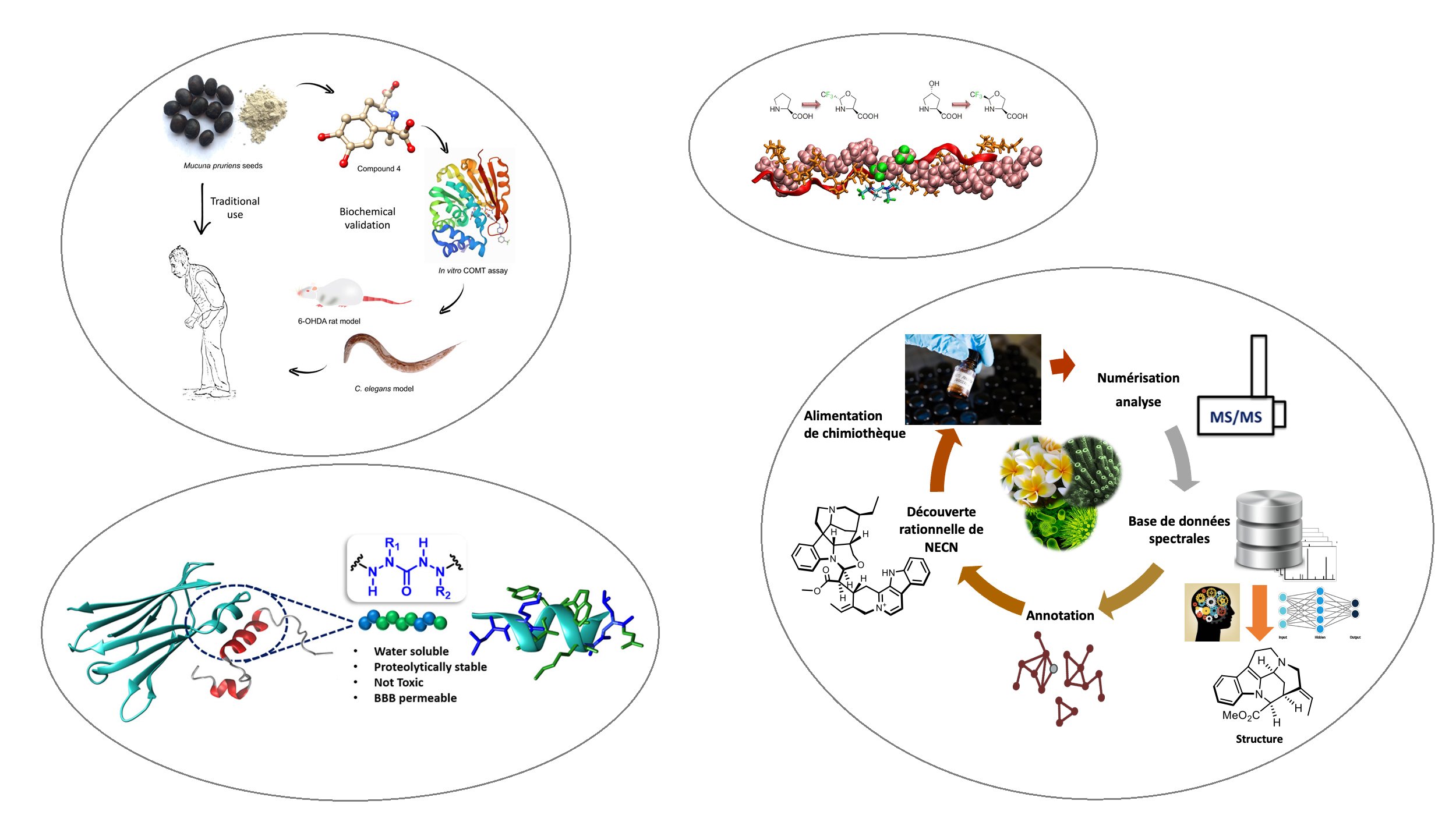
site en cours de contruction
Acteur majeur de la recherche académique au sein de l’UFR Pharmacie – Université Paris-Saclay, l’UMR CNRS BioCIS (Biomolécules : Conception, Isolement, Synthèse) perpétue une excellence dans les domaines de la synthèse organique et de la chimie médicinale en couvrant un large spectre d’expertises :
- en substances naturelles, comme source d’inspiration de molécules biologiquement actives ;
- en catalyse organométallique et le développement de méthodologies de synthèse (peptides & peptidomimétiques ; molécules fluorées, glycochimie, etc.)
- en modélisation moléculaire ; et
- en conception rationnelle de nouvelles entités chimiques (NECs) à visée thérapeutique et la compréhension de leur mode d’action.
Nos dernières actualités
Le TWEET de BioCIS – n° 23
TWEET - n° 23
Recrutement doctorant-e en chimie bioorganique
Rational design of fluorinated peptides for the inhibition of amyloid protein aggregation Grégory Chaume [...]
Recrutement Maitre de Conférences – Section 32
Recrutement d'un(e) Maitre de conférence. Enseignement : Le département Génie Biologique forme des spécialistes de laboratoires médicaux et industriels (parcours [...]
Recrutement ATER – Chimie – Biochimie
Poste ATER - Sections 32 - 64 Recrutement d'un(e) ATER pour l'année 2025-2026 Enseignement : Le/la Attaché(e) Temporaire d’Enseignement [...]
Le TWEET de BioCIS – n° 22
TWEET - n° 22
Félicitations Dr Boutahri
Toute l'équipe félicite Yazid Boutahri après sa soutenance de thèse et l'obtention du grade de Docteur de CY Cergy-Paris Université. [...]